
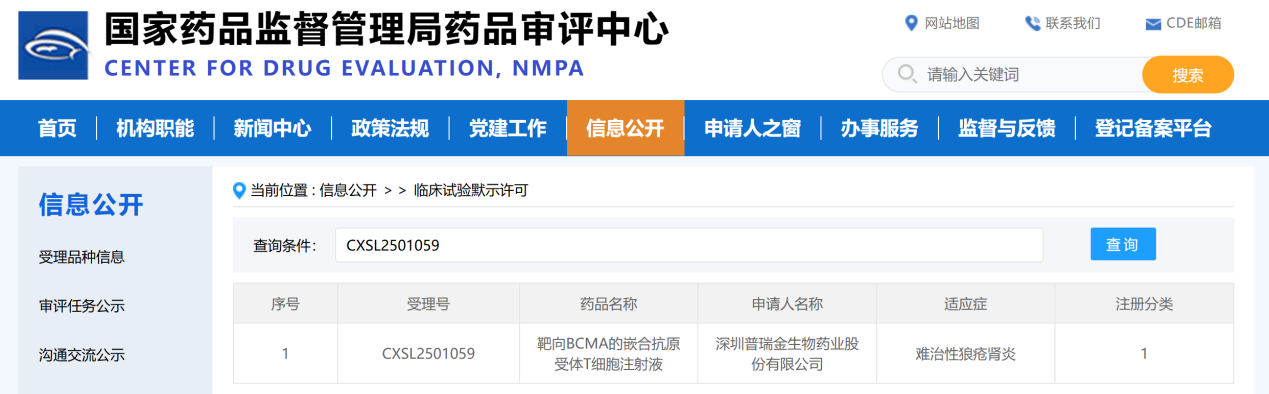
SHENZHEN, China, March 6, Shenzhen Pregene Bio-Pharmaceutical Co., Ltd. ("pregene") today announced that its independently developed BCMA-targeting Chimeric Antigen Receptor T Cell Injection has received Clinical Trial Approval from the National Medical Products Administration (NMPA) for a new indication. While the product progresses steadily for multiple myeloma, the Investigational New Drug (IND) application (Acceptance No.: CXSL2501059) for refractory lupus nephritis has been officially approved, authorizing the Company to initiate clinical trials for this indication.
About the BCMA-targeting CAR-T Cell Therapy
pregene's independently developed BCMA-targeting Chimeric Antigen Receptor T Cell Injection is a CAR-T cell therapy targeting BCMA, incorporating high-affinity and high-specificity heavy-chain variable domains. The product's research data, manufacturing process, and quality control systems have been developed in accordance with international standards, offering high production efficiency and significant cost advantages. The process and quality control systems demonstrate excellent reproducibility and stability.
About Lupus Nephritis
Lupus nephritis is a kidney disorder caused by systemic lupus erythematosus (SLE), characterized by lesions in glomerular podocytes, tubulointerstitium, or renal vessels, and is considered a severe condition. Among SLE patients, approximately 50% present with clinical manifestations of kidney damage, while renal histopathological examination reveals renal involvement in up to 90% of cases. In China, lupus nephritis is a common cause of end-stage renal disease (ESRD) and a significant contributor to mortality in SLE patients.
About pregene
Shenzhen Pregene Bio-Pharmaceutical Co., Ltd. is a National High-Tech Enterprise, a Guangdong Provincial Engineering Technology Research Center for Cell and Gene Therapy Innovative Drugs, and a Shenzhen "Specialized, Refined, Differential, and Innovative" Enterprise. Its shareholders include the National SME Development Fund, Casstar, Haier Capital, Shenzhen Capital Group, and Huapont Life Sciences (002004). Over the years, pregene has focused on cell and gene therapy, with a product pipeline encompassing multiple targets such as CAR-T, TCR-T, and CAR-NK therapies for a wide range of indications, including hematologic malignancies, solid tumors, and autoimmune diseases. Adhering to a development strategy of technology platformization, platform productization, product commercialization, and commercial internationalization, pregene stands as a leading innovative cell therapy company that has pioneered global multi-regional licensing of CAR-T cell therapies.
